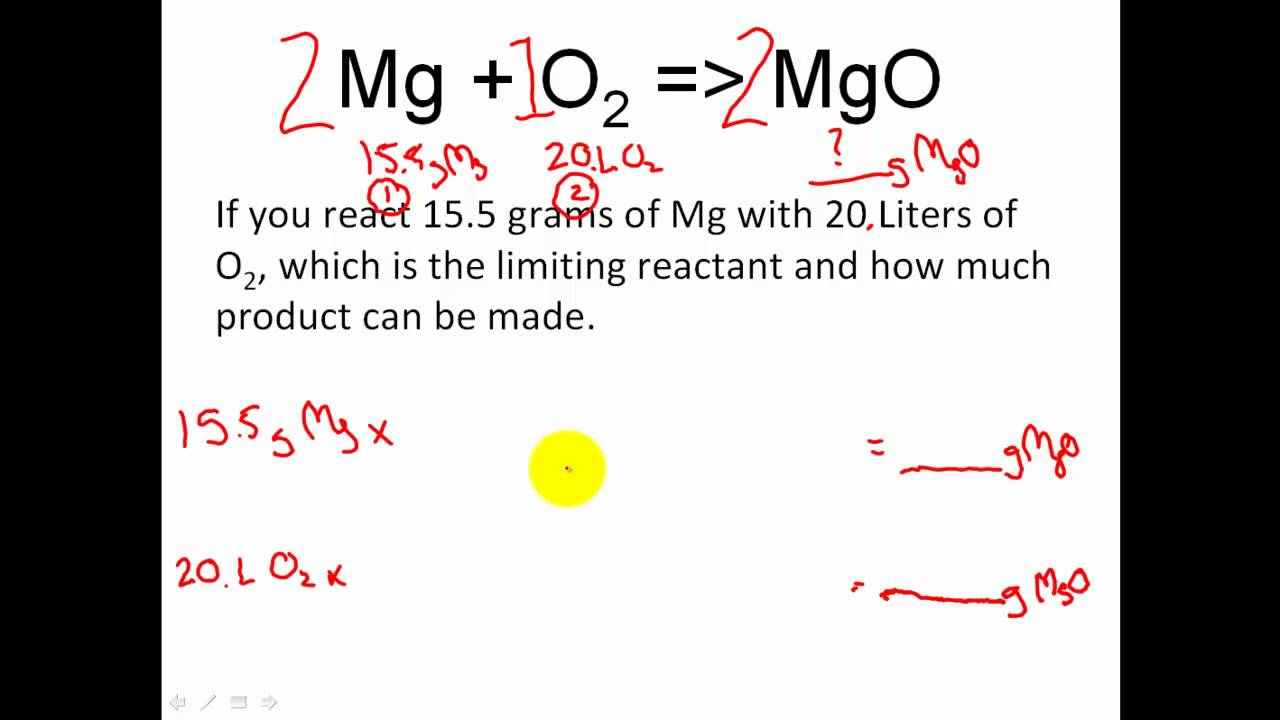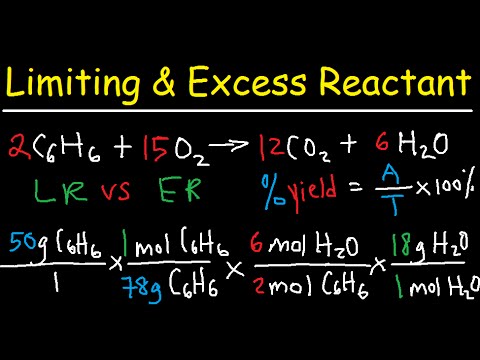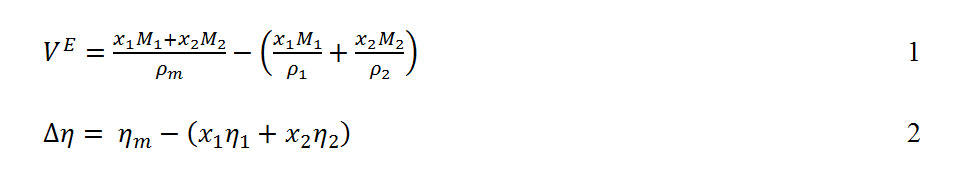
Derived thermodynamic properties of binary mixtures of m-Xylene, o-Xylene, and p-Xylene, with N,N-Dimethylformamide at T = (293.15, 303.15, 313.15 and 323.15) K : Oriental Journal of Chemistry

Chemical Equations and Reaction Stoichiometry. Chemical equations are used to describe chemical reactions, and they show (1)The substances that react, - ppt download

SciELO - Brasil - DENSITY, REFRACTIVE INDEX, APPARENT VOLUMES AND EXCESS MOLAR VOLUMES OF FOUR PROTIC IONIC LIQUIDS + WATER AT T=298.15 AND 323.15 K DENSITY, REFRACTIVE INDEX, APPARENT VOLUMES AND EXCESS
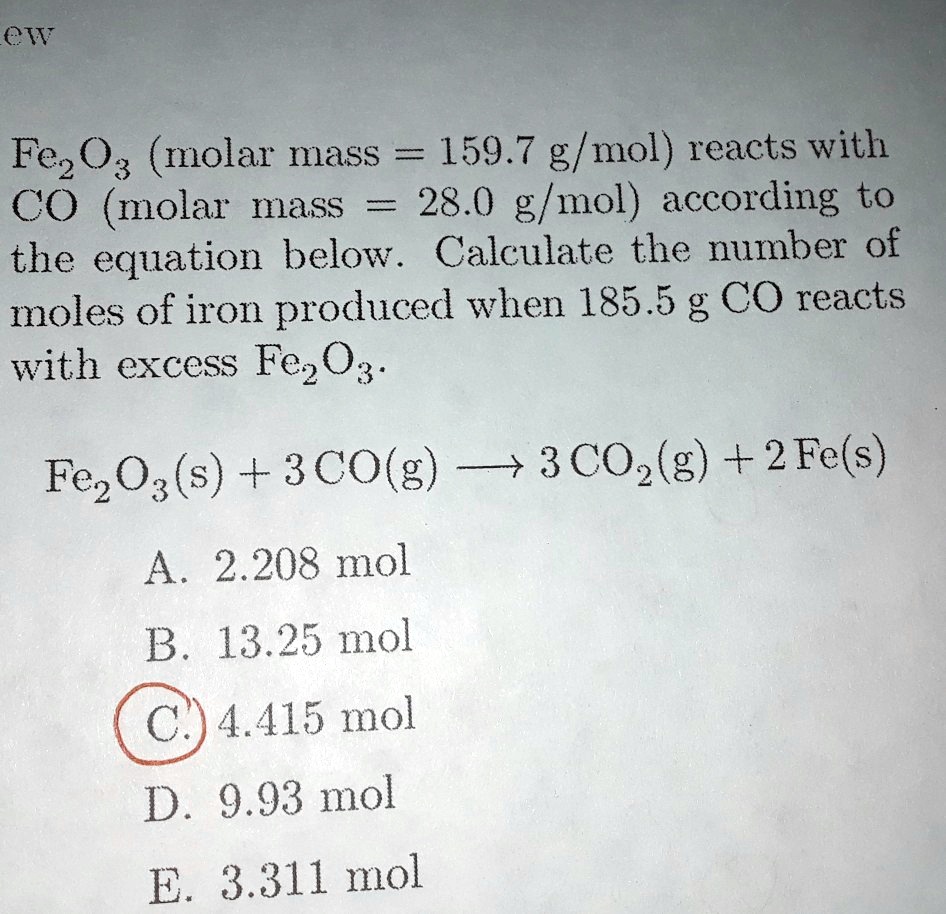
SOLVED: ew Fez O3 (molar mass = 159.7 g/mol) reacts with CO molar mass 28.0 g/mol) according to the equation below. Calculate the number of moles of iron produced when 185.5 g

200 mol of ethane are burned in a furnace with 50% excess air. A conversion of 95% is achieved. Calculate the composition of the stack gases?

Limiting Reactants & Excess. Limiting Reactant Calculations In many chemical reactions an excess of one reactant is added to ensure complete reaction. - ppt download
R ational Interpolation F orm ulae for the Excess Functions o f B inary M ix tu res H. Brodowsky Institut für Physikalische Che